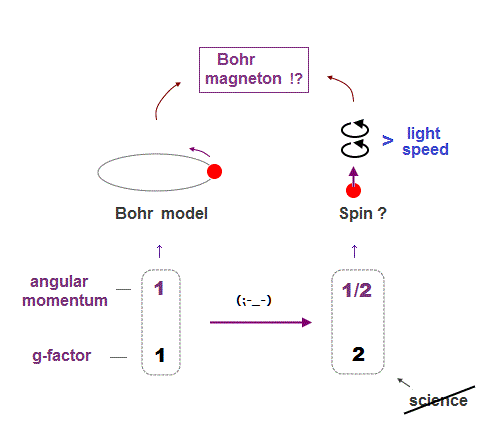
He took inspiration from the work of Planck and proposed that the atomic electrons are restricted to certain orbits Not continuously radiate energy, Bohr concluded that Maxwell's classical theory of electromagnetism does not apply onĪn atomic scale. When a friend suggested that Balmer's formula for the spectral lines of hydrogen might be relevant: "the whole thing was immediately clear toīohr's theory retains the classical idea that electrons move in circular orbits about a central nucleus. 
He became convinced that the new quantum theory of light might provide a clue to the understanding of atomic structure, and Manchester (England) and learned about the successes and problems associated with Rutherford's nuclear model of the atom. Niels Bohr spent most of his life in Copenhagen but after receiving his doctorate (in 1911) he visited Rutherford's laboratory in Where f represents the frequency of the light emitted and hf is the corresponding photon energy. So Eq.(2) can be rewritten in the more general form: This prediction has proven correct (see Table 3.1 and Fig. (IR) regions of the spectrum, corresponding to different values of the integer m. Balmer went on to speculate that other series of lines might exist in the ultraviolet (uv) and infrared Where C = 364.56 nm, m = 2 and the integer n takes values of 3, 4, 5 and 6 for lines in the visible region and higher values for further lines Wavelength positions of lines observed in the emission spectrum of hydrogen: Balmer (a Swiss schoolteacher) published a formula which accurately predicted (to better than 0.1%) the More recently, the elemental composition of many stars has been determinedīy attaching an optical spectrometer to an astronomical telescope. Passed through a flame containing sodium atoms (Fig. They occurred at the same wavelengths as the emission lines from a sodium source and were enhanced when the sunlight was 
In 1859, Gustav Kirchhoff showed that Fraunhofer's D-lines originated from the presence of sodium vapour in the sun's atmosphere Is lower and the atoms are further apart, such that they absorb certain wavelengths selectively, producing the dark Fraunhofer lines. To reach us, however, the radiation must pass through cooler layers of the sun's atmosphere, where the pressure 
Under these conditions, the brightĮmission lines broaden and overlap (due to Doppler and pressure broadening) to give a continuous wavelength distribution, similar to thatĮmitted by a heated solid. Originates from nuclear reactions in the sun's interior, which is at a high pressure and high temperature. We now know that the smooth background arises from the fact that the radiant energy There are many other lines in between see Fig. However, close observation of the spectrum of sunlight shows that it contains dark lines superimposed on a smooth background,Īs originally reported by Joseph Fraunhofer in 1814 (he labelled the prominent lines A, B, C, D etc., but modern spectroscopy shows that Was sufficiently developed that two new elements (cesium and rubidium) were discovered by analysing the light emitted by minerals which Wavelengths and relative intensities) is characteristic of the type of gas emitting the light (Fig. Through a spectrometer, containing a glass prism or a diffraction grating as the light-dispersing element. Such line spectra can be observed by passing the light from a gas-discharge lamp Of a hot gas, however, whose atoms are much farther apart from each other than in a solid, the emission spectrum consists of a series ofīright lines superimposed on a dark background. We have seen that the spectrum of electromagnetic radiation emittedīy a hot solid contains of a continuous range of wavelengths, whose intensity distribution is given by Planck's formula. Optical Spectroscopy and Bohr's Model of the Atomīefore discussing Niels Bohr's contribution to our understanding of the atom, we need to go back to the 19th century andĬonsider developments in the science of optical spectroscopy. Figure references are to the second edition of Modern Physics by Serway, Moses and Moyer (Saunders, 1989).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
